|
11/6/2023 0 Comments Net dipole moment
Hence, favourable factors for ionic bond formation are as follows: Bond formation also depends upon the lattice energy of the compound formed. Hence, the formation of ionic bonds depends upon the ease with which neutral atoms can lose or gain electrons. Write the favourable factors for the formation of ionic bond.Īn ionic bond is formed by the transfer of one or more electrons from one atom to another. (e) This rule cannot be applied to those compounds in which the number of electrons surrounding the central atom is less than eight. For example, NO and NO 2 do not satisfy the octet rule. 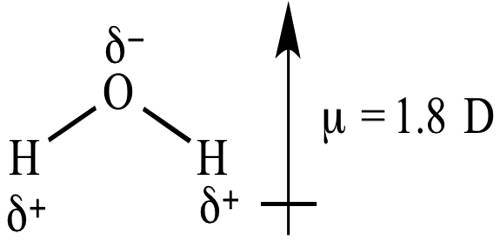
(d) The octet rule is not satisfied for all atoms in a molecule having an odd number of electrons. The elements present in these periods have more than eight valence electrons around the central atom. (c) The octet rule cannot be applied to the elements in and beyond the third period of the periodic table. However, some noble gases like xenon and krypton form compounds such as XeF2, KrF2 etc. (b) It is based upon the inert nature of noble gases. (a) The rule failed to predict the shape and relative stability of molecules. The following are the limitations of the octet rule: The octet rule successfully explained the formation of chemical bonds depending upon the nature of the element. According to this rule, atoms can combine either by transfer of valence electrons from one atom to another or by sharing their valence electrons in order to attain the nearest noble gas configuration by having an octet in their valence shell. The octet rule or the electronic theory of chemical bonding was developed by Kossel and Lewis. Draw the Lewis structures for the following molecules and ions:ĥ. 
The uninegative charge infers that there will be one electron more in addition to the one valence electron. The Lewis dot symbol of hydrogen (H) is H. The number of valence electrons in hydrogen is 1. The tripositive charge on a species infers that it has donated its three electrons. 
The Lewis dot symbol of aluminium (Al) is. The number of valence electrons in aluminium is 3. The dinegative charge infers that there will be two electrons more in addition to the six valence electrons. The number of valence electrons in sulphur is 6. Write Lewis symbols for the following atoms and ions: Hence, the Lewis dot structure is:īr: There are seven valence electrons in bromine. N: There are five valence electrons in an atom of nitrogen. O: There are six valence electrons in an atom of oxygen. Hence, the Lewis dot structure is:ī: There are 3 valence electrons in Boron atom. Na: There is only one valence electron in an atom of sodium. Mg: There are two valence electrons in Mg atom. Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br. An ionic bond is formed as a result of the transference of electrons from one atom to another.Ģ. The chemical bond formed as a result of sharing of electrons between atoms is called a covalent bond. This combination can occur either by sharing of electrons or by transferring one or more electrons from one atom to another. Atoms, therefore, combine with each other and complete their respective octets or duplets to attain the stable configuration of the nearest noble gases. Hence, it was postulated that the elements having incomplete outermost shells are unstable (reactive). It was observed that the inertness of noble gases was because of their fully filled outermost orbitals. Various theories have been suggested for the formation of chemical bonds such as the electronic theory, valence shell electron pair repulsion theory, valence bond theory, and molecular orbital theory.Ī chemical bond formation is attributed to the tendency of a system to attain stability. Explain the formation of a chemical bond.Ī chemical bond is defined as an attractive force that holds the constituents (atoms, ions etc.) together in a chemical species. Our Physics Wallah team Prepared Other Subjects NCERT Solutions for class 11.Ĭhapter 4 Chemical Bonding and Molecular Structureġ. NCERT Solutions for class-11 Chemistry Chapter 4 Chemical Bonding and Molecular Structure is prepared by our senior and renown teachers of Physics Wallah primary focus while solving these questions of class-11 in NCERT textbook, also do read theory of this Chapter 4 Chemical Bonding and Molecular Structure while going before solving the NCERT questions. \)).NCERT Solutions For Class 11 Chemistry chapter 4-Chemical Bonding And Molecular Structure
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
